 
Δ S is the entropy change of the system measured in J/K, and T – the temperature in Kelvin Rearranging the equation, we get an expression for Δ S: 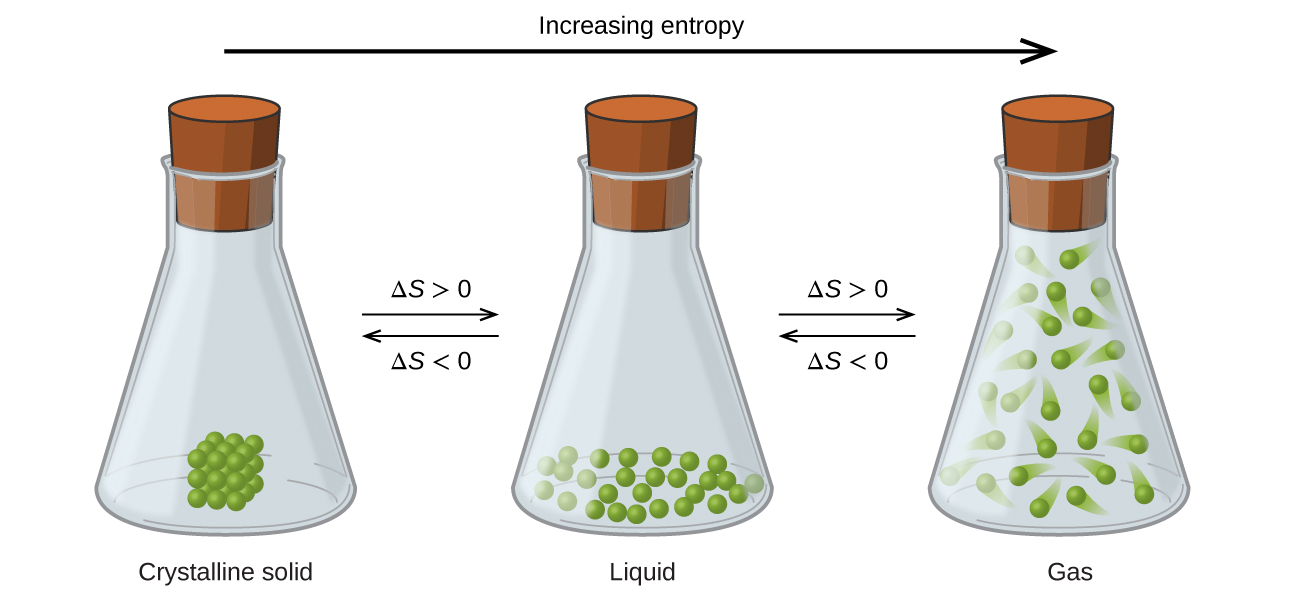
Remember, also that when the system is at equilibrium the Gibbs free energy is equal to zero: To derive an equation for the entropy change associated with phase transitions, we need to remember that these changes occur when the system is at equilibrium. Remember this pattern, and the corresponding terms for each pair of opposite processes: melting/fusion vs freezing, evaporation/vaporization vs condensation.Ĭalculating the Entropy Change of State Changes These are the points of phase transitions where, for example, the liquid turns into a gas even at the same temperature. Notice how the entropy is still increasing in the regions where the temperature is not changing. The phase transition graph showing the entropy vs temperature is very useful to visualize this concept: This behavior is explained by the increasing freedom of motion when molecules go from the most ordered solid state to liquid, and then a gas state where the degree of randomness is the highest. On the basis of determining the entropy change associated with phase transitions is the third law of thermodynamics: the entropy of a perfect crystalline substance is zero at the absolute zero temperature.Īn implication of this is that the entropy is the lowest in solids, and it keeps increasing in the order of going to liquid and gas states: solid < liquid < gas. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
